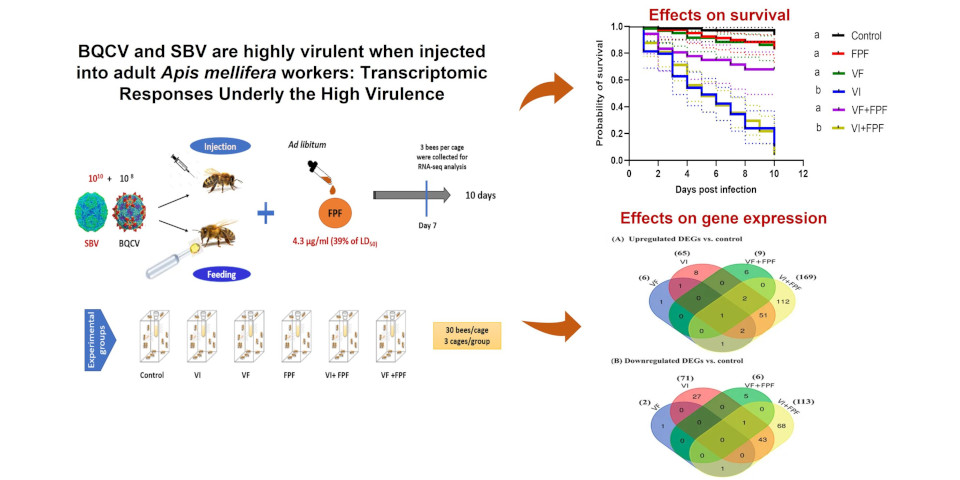
Background: Over the last two decades, honey bees (Apis mellifera) have suffered high rates of colony losses that have been attributed to a variety of factors, chief among which are viral pathogens, such as deformed wing virus (DWV), whose virulence has increased because of vector-based transmission by the invasive, ectoparasitic varroa mite (Varroa destructor). A shift in the experimental mode of transmission of the black queen cell virus (BQCV) and sacbrood virus (SBV) from fecal/food–oral (direct horizontal) to vector-mediated (indirect horizontal) transmission also results in high virulence and viral titers in pupal and adult honey bees. Agricultural pesticides represent another factor that acts independently or in interaction with pathogens, and they are also thought to cause colony loss. Understanding the molecular mechanisms underlying the higher virulence following a vector-based mode of transmission provides deeper insight into honey bee colony losses, as does determining whether or not host–pathogen interactions are modulated by exposure to pesticides. Methods: Through an experimental design with controlled laboratory, we investigated the effects of the modes of transmission of BQCV and SBV (feeding vs. vector-mediated via injection) alone or in combination with chronic exposure to sublethal and field-realistic concentrations of flupyradifurone (FPF), a novel agricultural insecticide, on honey bee survival and transcription responses by using high-throughput RNA sequencing (RNA-seq) analysis. Results: Co-exposure to viruses via feeding (VF) or injection (VI) and FPF insecticide had no statistically significant interactive effect on their survival compared to, respectively, VF or VI treatments alone. Transcriptomic analysis revealed a distinct difference in the gene expression profiles of bees inoculated with viruses via injection (VI) and exposed to FPF insecticide (VI+FPF). The number of differentially expressed genes (DEGs) at log2 (fold-change) > 2.0 in VI bees (136 genes) or/and VI+FPF insecticide (282 genes) was very high compared to that of VF bees (8 genes) or the VF+FPF insecticide treatment (15 genes). Of these DEGs, the expression in VI and VI+FPF bees of some immune-related genes, such as those for antimicrobial peptides, Ago2, and Dicer, was induced. In short, several genes encoding odorant binding proteins, chemosensory proteins, odor receptors, honey bee venom peptides, and vitellogenin were downregulated in VI and VI+FPF bees. Conclusions: Given the importance of these suppressed genes in honey bees’ innate immunity, eicosanoid biosynthesis, and olfactory associative function, their inhibition because of the change in the mode of infection with BQCV and SBV to vector-mediated transmission (injection into haemocoel) could explain the high virulence observed in these viruses when they were experimentally injected into hosts. These changes may help explain why other viruses, such as DWV, represent such a threat to colony survival when transmitted by varroa mites.
2447 Members
127 Countries!
127 Countries!